

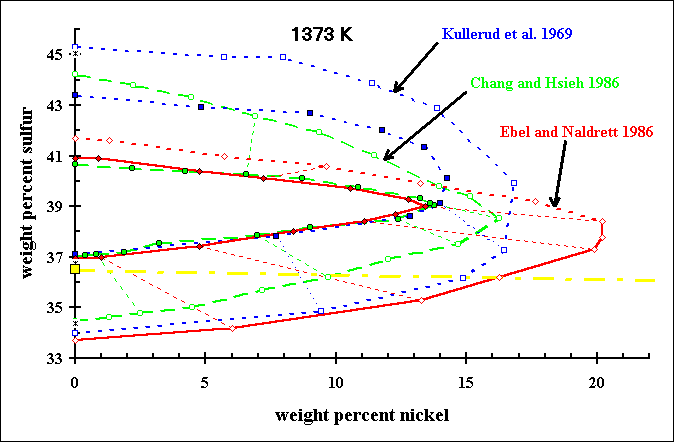
 Comparison of MSS-Liquid Tielines in Fe-Ni-S at 1373K...Kullerud et al. (1969) analysis of XRD patterns of experiments ...Chang and Hsieh (1986) calculations using thermodynamic model ...Ebel and Naldrett (1996) microprobe analysis of experiments The join between FeS (troilite, yellow square) and NiS (millerite) is illustrated in yellow for reference. |
The stability field of MSS, and tie-lines to coexisting sulfide liquid, at 1373 K
are illustrated here. The data for Kullerud et al. (1969) and Chang and Hsieh(1986) were read from their published figures.
Sulfur contents in the Ebel and Naldrett (199) experimental liquids at high sulfur content are minimum bounds, because such
liquids do not retain sulfur when quenched from high temperature. Our new data on the partitioning of Ni between MSS and liquid establish the first accurate partition coefficients to be determined experimentally for this system. Similar data have been obtained for Cu.
|
||||||
| ----------------------------- | ----------------------------- | ||||||
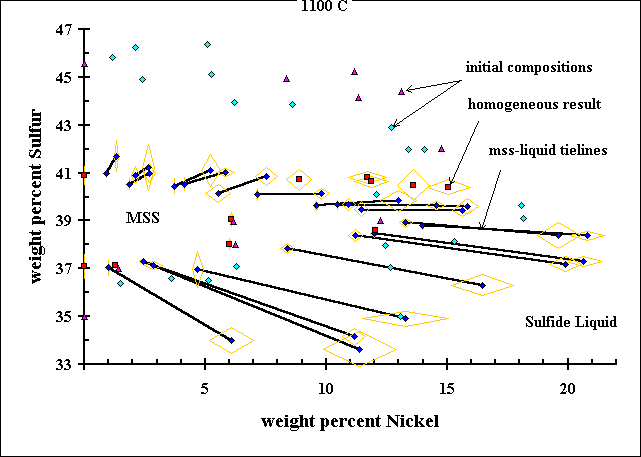 Experimental Results at 1373 KAll the results are averages of many data points on each phase, which have the standard deviation shown by the large gold diamonds around each point. All the compositions have been normalized to total 100wt%. |
This figure illustrates the experimental data of Ebel and Naldrett (1969) in greater detail. The tie-lines between MSS and Sulfide Liquid are revealed by the compositions of coexisting material in the experiments. Some bulk compositions in the interior of the MSS field were run to make sure the technique works properly, and the results (homogeneous MSS) show the accuracy of the microprobe technique and the reliability of the experimental method. Some experiments were run in duplicate, and they can be seen to yield results which are identical within one standard deviation of the microprobe results. ----------------------------- |
----------------------------- |
|
 Detail of High Sulfur Experiments
Some experiments on the sulfur-rich side of the system, starting above 41 wt% S, yielded homogeneous sulfide liquid,
but this liquid was missing sulfur. The reason for this is that sulfur forms vapor at the temperature of the
experiment, and this vapor condenses on the wall of the tube when the experiment is quenched. Furthermore, some of the
sulfur in the liquid at high temperature probably escapes the liquid as it is being quenched, regardless of how fast it
is quenched. This sulfur loss is not a problem for experiments with less than about 39 wt% S,
and it can be seen that the bulk compositions of these experiments lie very close to the tielines joining the results. |
----------------------------- |
----------------------------- |
|